 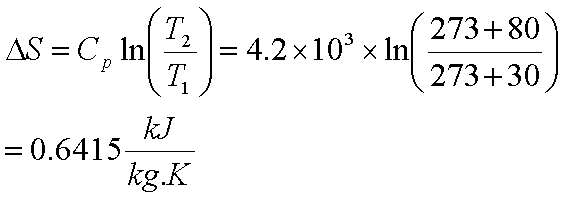
melting, evaporation) or chemical change (e.g. Reactions proceed spontaneously in the direction that lowers their Gibb’s free-energy:Ī reaction is deemed spontaneous (feasible), if ∆G0 : known as endergonic reactionĭefining entropy and determining entropy changes from absolute entropy valuesĮntropy change, ∆S, relates to increasing disorder of a process, either arising through physical change (e.g. The feasibility of a physical or a chemical change depends on the balance between the thermodynamic quantities of enthalpy change (Δ H), entropy change (Δ S) and temperature ( T). 298K) ΔG ⦵ Gibbs free-energy change, measured in kJ mol −1 ΔH ⦵ Enthalpy change, measured in kJ mol −1 T Temperature measured in Kelvin ΔS ⦵ Entropy change, measured in J K −1 mol −1 ⦵ Standard pressure (100kPa), usually with a stated temperature (e.g. Classification, variation, food webs and pyramids.Combustion reactions and impact on climate.Atoms elements compounds and mixtures (interactive).B1.6 Waste materials from plants and animals.RSC Learn Chemistry Classic Chemistry Experiments.RSC Classic Chemistry Experiments (1995).Practical Chemistry (Nuffield Foundation/RSC).3.15 Nuclear magnetic resonance spectroscopy.3.14 Organic synthesis and analysis (A2).2.6 Reactions of ions in aqueous solution.2.4 Properties of Period 3 elements and their oxides.1.11 Electrode potentials and electrochemical cells (Redox A2).1.10 Equilibrium constant Kc for homogeneous systems (Equilibrium A2).3.6 Organic analysis (AS): analytical techniques.1.7 Oxidation reduction equations (Redox AS).1.6 Chemical equilibria and Le Chatelier’s principle.C3.5 Production of ammonia (an example of a reversible reaction). 
C3.4 Further analysis and quantitative chemistry.C3.3 Calculating and explaining energy change.C2.5 Exothermic and endothermic reactions.C2.3 Atomic structure, analysis and quantitative chemistry.C1.7 Changes in the Earth and its atmopshere.C1.5 Other useful substances from crude oil.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
